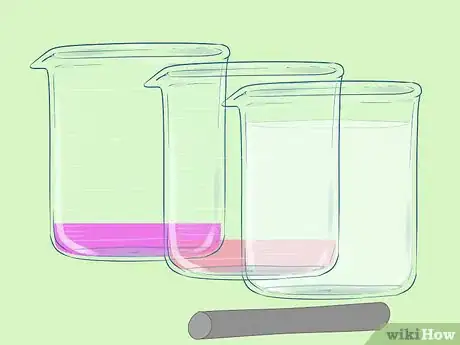X
wikiHow is a “wiki,” similar to Wikipedia, which means that many of our articles are co-written by multiple authors. To create this article, volunteer authors worked to edit and improve it over time.
This article has been viewed 25,294 times.
Learn more...
Everybody knows how you can make a battery from a lemon. You can also make batteries from cola or salt water. The problem is, these batteries have a low voltage. You can make a high-voltage battery using electrochemistry.
Steps
Part 1
Part 1 of 5:Getting the Materials
Part 1
-
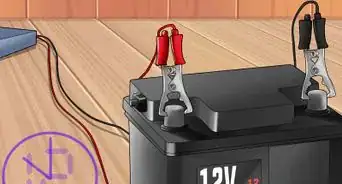
1Gather your materials. You will need: two glass beakers, filter paper and potassium nitrate for the salt bridge, aluminium foil and one molar aluminium nitrate solution for the negative electrode, wires and crocodile clips. The materials for the positive electrode vary. You will also need something to act as a circuit load, like a light bulb.
- One molar = one mole per litre.
Part 2
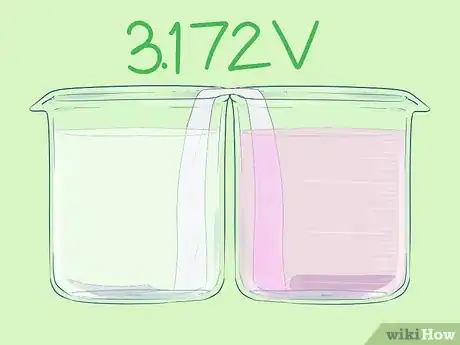
Part 2 of 5:Preparing the Salt Bridge
Part 2
Part 3
Part 3 of 5:Preparing the Negative Half-Cell
Part 3
-
1Add the aluminium nitrate solution to one of the beakers.
-
2Place a strip of aluminium foil into the solution. The strip should touch the bottom of the beaker. It should not touch the salt bridge. You may wish to bend the top of the strip over the edge of the beaker. This aluminium strip is the electrode.
- You have formed an Al3+/Al half cell.
Part 4
Part 4 of 5:Preparing the Positive Half Cell
Part 4
Using Cu2+/Cu
-
1Gather your supplies. You will need: a strip of copper and some one molar copper nitrate solution.
-
2Add the copper nitrate solution to the second beaker.
-
3Place the copper strip in the second beaker. It should touch the bottom, but not the salt bridge. This is the positive electrode.
- This cell has a voltage of 1.821V.
Using Fe3+/Fe2+
-
1Gather your supplies. You will need: one molar iron (III) nitrate (Fe(NO3)3) solution, one molar iron (II) nitrate (Fe(NO3)2) solution, and a conductive graphite rod.
-
2Add equal amounts of each of the iron nitrates to the second beaker.
-
3Place the graphite rod into the beaker. It should not touch the salt bridge. This is the positive electrode.
- This cell has a voltage of 2.432V.
Using Cr2O7 2- + 14H+/Cr3+
-
1Get the supplies for this part. You will need: one molar potassium dichromate solution, one molar nitric acid, one molar chromium nitrate solution, and a conductive graphite rod.
-
2To the second beaker, add: one measure of potassium dichromate solution, two measures of chromium nitrate solution, and an excess (>14 measures) of nitric acid. One measure should be the largest amount that you can add without the beaker overflowing after everything is added.
-
3Add the graphite rod. It should not touch the salt bridge. This is the positive electrode.
- This cell has a voltage of 2.992V.
Using MnO4- + 8H+/Mn2+
-
1Gather the correct supplies. You will need: one molar potassium permanganate solution, one molar nitric acid, one molar manganese nitrate solution, and a conductive graphite rod.
-
2To the second beaker, add: one measure of potassium permanganate solution, one measure of manganese nitrate, and an excess (>8 measures) of nitric acid. One measure should be the largest amount that you can add without the beaker overflowing after everything is added.
-
3Add the graphite rod. It should not touch the salt bridge. This is the positive electrode.
- This cell has a voltage of 3.172V.
Part 5
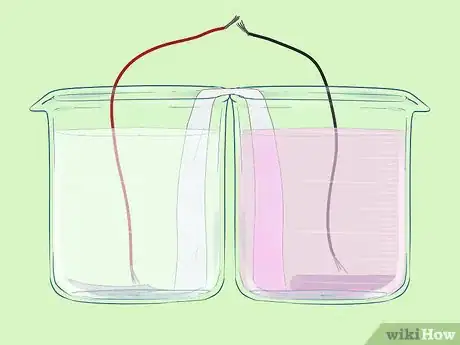
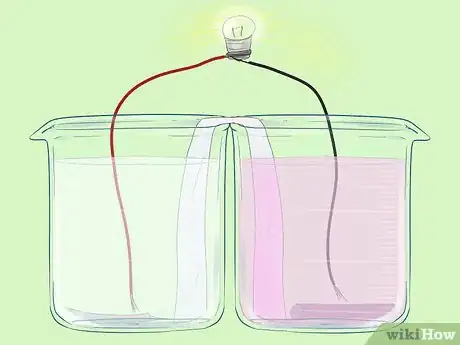
Part 5 of 5:Completing the Circuit
Part 5
Warnings
- Assume all chemicals are corrosive, toxic and flammable. Take appropriate precautions.Thanks!